BioNTech SE – “One hit wonder” or Game changing Biotech platform ?
This is not investment advice. PLEASE DO YOUR OWN RESEARCH !!!
Extra Health Warning
I guess some old time readers will think: Now that guy really lost his marbles or so. I have very little knowledge about Biotech companies and the industry in general and nothing qualifies me to write about a Biotech stock. My only experience with Biotech companies during the 10 years of the blog was Swiss based Actelion, but only as a special situation with a Spin-off component. So you might take this post as a warning signal that markets have become overheated and the author of this blog has indeed lost his marbles.
Management Summary:
Despite my limited experience and understanding of the sector, I do think that BioNTech represents an interesting “bet” on the success of the underlying mRNA technology and the ability of BioNTech to establish (or having already succeeded) a platform that will yield much more than just this one blockbuster Covid-19 vaccine but many other successful vaccines and cancer drugs (and more). Their intention to become a “full fledged” pharmaceutical company could be the start of a long “Compounding story” if successful, but there are also clearly many risks involved.
BionTech history IPO & “Pivot”
Up until a few months ago, BioNTech was “just another Biotech company” that IPOed in September 2019 in the US. Since a few months however, BioNTech has become almost a household name all over the world, as they have developed the first and so far most effective vaccine against Covid-19.
It is very interesting to read the F-1 IPO prospectus of BioNTech. Back then, their product pipeline was 100% geared towards cancer and this were the main risk factors:
Our business is subject to a number of risks of which you should be aware before making an investment decision. These risks are discussed more fully in the section of this prospectus titled “Risk Factors” immediately following this prospectus summary. These risks include, but are not limited to, the following:
• We are a clinical-stage biopharmaceutical company with no pharmaceutical products approved for commercial sale.
• We have incurred significant losses since our inception and we anticipate that we will continue to incur significant losses for the foreseeable future.
• We will require substantial additional financing to achieve our goals.
• We will need to develop and expand our company, and we may encounter difficulties in managing this development and expansion, which could disrupt our operations.
• Pharmaceutical product development is inherently uncertain, and there is no guarantee that any of our product candidates will receive marketing approval.
• No mRNA immunotherapy has been approved, and none may ever be approved, in this new potential category of therapeutics. mRNA drug development has substantial clinical development and regulatory risks due to the novel and unprecedented nature of this new category of therapeutics.
So they had so far no pharmaceutical product that was approved and not even a single mRNA treatment had ever been approved back then.
According to several stories, the BioNTech founder and CEO Ugur Sahin started extremely early to “gamble” his company on the vaccine as it is mentioned here in the FT:
Two months before the World Health Organization had even declared a pandemic, Dr Sahin suggested that the 12-year-old firm devote the vast bulk of its meagre resources to coming up with a vaccine. “He has a very high hit rate when it comes to predicting outcomes,” Dr Tureci says of her husband — a trait she jokingly admits was initially “annoying”. She and the rest of the company’s board approved the plan soon after.
Clearly some luck was at play here as well, but very often, luck hits those who are prepared.
Stock price/ Valuation: “One hit wonder” vs. “Platform”
Looking at the stock price, it is pretty clear that I am unfortunately not the first one who has noticed the success of BionTech: From the IPO price of 1 USD, the stock has increased to more than 6x its valuation back in September 2019 and clearly some shareholders are very happy at a market cap of ~23 bn EUR:
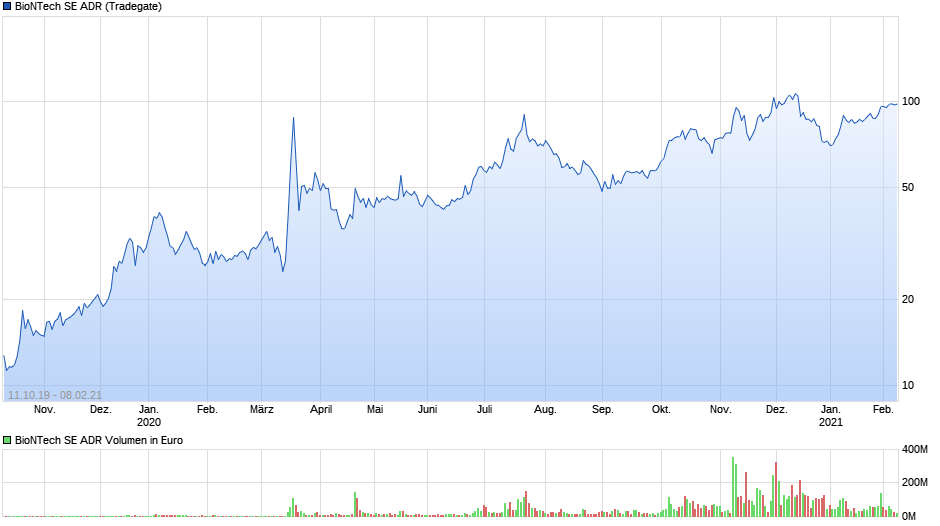
According to what I have found on the internet, BioNTech and Pfizer are selling the vaccine at around 15 USD per shot and that the original deal was a revenue split of 50/50. It currently looks like that so far ~ 1bn doses have been ordered and that for 2022 there are some news articles that up to 3 bn doses could be required.
So Even if we optimistically assume that BioNTech could actually sell 5 bn doses in the next 3-5 years or so, this will “only” generate up to 35 bn EUR total sales and maybe 10 billion (?) profits after tax and would not justify the current valuation. This is where the underlying mRNA technology enters the picture.
mRNA technology as a platform
In the past, pharmaceutical companies more or less were chemical companies, extracting and mixing together some ingredients and sold them as therapeutics.
Biotech companies “disrupted” this field by actually using genetically modified bacteria to create more complex molecules that first replaced existing (animal) substances (Insulin) before moving to new substances that could be used as treatments where “chemical” pills did not succeed.
mRNA technology as far as I understand it is now one step further: mRNA therapeutics are also produced in a “biotech” type of way, but instead of producing a finished product, mRNA “medicine” stimulates the body’s own cells to create proteins which then again can fight illnesses within the body (many MRNA links at the end of this post).
BioNTech and other mRNA players claim that this is a new kind of platform that enables them to fight different diseases, such as cancer or other hard to battle illnesses as their way leads to much more targeted results.
These days every company wants to be a run “platform” so I am very skeptical on any company claiming to be the next platform. However in BioNTechs case with mRNA, I think there could be some truth in that claim. The fact that they developed a new vaccine not only faster than the existing specialists but also with a superior outcome could be luck, but maybe also the result of actually having established a kind of revolutionary drug development platform. in addition, BioNTechs vaccine seems to be easier to manufacture if you know how to do it.
The obvious next step would be clearly to develop more (and better) Vaccines for instance against the flu. According to sources, the flu vaccine market alone is around 5 bn USD sales per year and this is based on pretty ineffective vaccines. If BionTech would be able to develop a more efficient and easier to manufacture flu vaccine, I think it is not unrealistic that they could both, expand the category and and gain significant market share.
A quick back of the envelope calculation (assuming 25% EBIT margins) would require around 4 bn in annual vaccine sales for BioNTech (on top of the one time Covid-19 vaccine sale) to more or less justify today’s valuation. This would be less than 10% of the current global vaccine market which I don’t find unrealistic as mRNA vaccines seem not only to be more efficient but also quicker to manufacture.
If they would really be successful to develop cancer treatments, the valuation clearly has significant upside in my opinion, but I have no clue how much. The biggest blog buster drugs seem to have generated sales over 100 bn USD in their lifetime, However I have no idea how valuable BioNTechs current pipeline is at the moment
In the Spiegel interview, Sahin and Türeci mention a few interesting things such as:
DER SPIEGEL: BioNTech’s origins are actually in cancer research. You first attracted attention three years ago with a successful study on a vaccine against cancer. When will the first drug of that type be approved?
Türeci: We have several products in the pipeline, and major efficacy studies for some of them are approaching. In other words: We believe it could be 2023/24.
Şahin: At the moment, we are mainly working in two areas: infectious diseases and cancer research. But we also want to develop beyond these core areas. In the medium term, we see ourselves as an immunotherapy company. We will be addressing inflammation, autoimmune diseases and regenerative therapies. There will surely be some product candidates in those areas in the next year or two.
DER SPIEGEL: Progress has accelerated particularly rapidly recently in the area of oncology. Some experts are hopeful that, at this rate, many forms of cancer will be readily treatable by the end of the decade. Are you as optimistic?
Şahin: We are on the path to detecting and treating cancer earlier. This could be a real revolution. What is still missing are curative procedures for early-stage patients. Thirty to 40 percent develop metastases again at some point after surgery. This is an area in which we will be particularly active with BioNTech. And I see a realistic chance that we will succeed in fundamentally changing how cancer is treated.
My current impression is that these guys are no bullshitters but know what they are talking about and they have proven themselves already multiple times. Money is not a problem for many years and with the vaccine success, they might have it easier going forward.
CEO/Founder/Shareholders/ other VIPs
As I mentioned in the blog earlier, the founders, a married couple are children of Turkish immigrants. Interestingly, BioNTech is their second company. The first company, Ganymed, that specialized in more “traditional oncology drugs, became a “Unicorn” when it was sold in 2016 to Japanese company Astellas for 1.2 bn EUR (including earn outs). It seems that the wife ran Ganymed as CEO and in parallel, Ugur founded BioNTech in 2008 to focus on more “cutting edge” technology.
There is an interesting 8 year old video (German) about the couple and their activities back then:
(Observation: Ugur really aged since then, but the vision of the platform was already there….). This is Ugur in Decmeber 2020:
Personally I do really like the way he communicates: very precise, understandable and no hype.
Another parallel to Ganymed is that the biggest investors were the Strüngemann brothers, two German self made billionaires who created Hexal, a leading German generics company that they sold themselves for around 5,6 bn EUR to Novartis in 2002. The combination of researchers with experienced pharma entrepreneurs seems to work. The Strüngemann brothers still own more than 50% of BioNtech at the time of writing. I guess this also helped them to strike co-operations and scale up production surprisingly fast.
One very important person with regard to the underlying mRNA technology seems to be Hungarian born Katalin Kariko who is one of the pioneers of mRNA technology and joined BioNTech in 2013. She seems to be one of the prime candidates for a Noble Prize in the next years.
On becoming a “full pharma” company
On several occasions, Sahin and also Strüngemann mentioned that they plan to create a new “full fledged pharma” company. Sahin’s motivation is relatively simple: He wants to bring treatments to patients, which he cannot ensure if he is just a researcher. The Strüngemann brothers on the other side want to prove that they cannot only create a pharma company that copies existing non-patented stuff (Hexal) but one that really creates new groundbreaking treatments.
This combination has worked well so far. I think it is also important that no one should expect that they will pay out big dividends. I think that they will reinvest most of the vaccine profits into building this business.
I think this is a risky but potentially smart move:
- there is a lot of political tailwind for a German/European “Champion”. The recent discussions have clearly shown the advantages of having supply chains and production in one’s own country.
- Although the vaccine is often named the “Pfizer vaccine”, BioNTech has become a brand name in a very short time in the relevant places. This has been a multi billion marketing campaign for free.
- With the exception of Moderna, BioNTech is currently building a significant and expanding competiitve advantage against other mRNA players: They learn how to mass produce mRNA vaccines and literally hundred of million people will hopefully verify that their drug is both safe and efficient. Why gamble on a competitor if you can have the “gold standard” ?
- I think the Strüngemann brothers have enough experience how to scale a pharmaceutical company which should limit the risk of this expansion
Risks
I mentioned above that some of the IPO risks have been resolved by the Covid-19 vaccine. However many risks remain, only to mention a few:
- mRNA competition (Moderna), especially in the short term as the BioNTec vaccine needs to be stored at -70 degrees celsius
- some problems down the road with the vaccine (long term adverse effects ?)
- political interference (Vaccines for free etc.)
- corporate espionage / Government sponsored (I am sure the Chinese or Russians try everything to get the “secret sauce”)
- Litigation law suits
- Key woman>/man risk
- “business model” risks from going full pharma
And I am sure there are another hundred risks that I didn’t mention or even don’t know about. Make no mistake: This is a risky company.
Pros/Cons
As always, a quick summary of pros and cons:
Pros:
- Good combination of entrepreneurial CEO and long term shareholders
- pandemic has accelerated development by at least 5-10 years
- clear competitive advantages in application of the technology, which increases due to mass production
- superior vaccine (easier to produce, more better efficacy)
- Interesting strategy to source in production
- political tailwind to produce “national champion”
- Pandemic was a free “marketing” event worth billions in sales and marketing euros
- underlying technology could indeed be a game changer, not only in the vaccine segment
- very positive short term results to be expected
Cons
- political uncertainty around vaccines
- application to other areas still unproven
- competition through other mRNA players (Moderna)
- Theft of intellectual capital
- competition through other, cheaper vaccines
- some residual risks of longer term side effect
- Key person risks
- “Double pivot” risk
- hard to value
Summary/game plan:
I have to admit that some “fear of regret” plays a role here. i would deeply regret not to have invested if this becomes an even bigger success. Therefore I decided to invest “only” 2% of the portfolio into BioNTech within my “Long term growth bucket” at around 98 EUR/share, reflecting the inherent risk at this stage.
My game plan would be to hold this at least 2-3 years and only consider to sell if the stock drops for no reason by more than -50% or goes up by more than +100%.
I expect very positive results both, for 2021 and 2022, however after than, vaccine sales will clearly go down and then it needs to be seen how the cancer pipeline works out or if they can come up with more vaccines.
If initial pipeline would become more concrete and the valuation still makes sense, I might increase the position in 2 or 3 years from now. In between, the position will motivate me to learn more about mRNA and Biotech in general, which is a nice side effect.
Another side effect is that theoretically, the stock could hedge me against a “Covid-19 worst case mutation” scenario for my more Tourism related stocks (Sixt, Wetherspoon) as more mutations would mean more business for BioNTech. this is however not the primary motivation.
I aslo need to manage overall risk in the protfolio. With Play Magnus, JET and BioNTech, I have now 3 high risk growth companies in the portfolio. The total exposure here is 7%. Anything above 10% would make me nervous.
________________________________________________________________________________________________________________________________
Links:
Interview with Sahin and his wife in Spiegel from December 2020
Good background story on mRNA, BioNTech, Moderna, Kariko etc.
Pre Covdi-19 comparison Moderna, BioNTech & Curevac (German)
I have waited for a long time, but I started to sell some BioNTech shares today. 1/10 of the initial position for EUR 271.
Sold another 1/10 of the original position at 290 EUR/share. Very likely too early. 80% of the initial position remains.
Sold another 1/10 at 300 EUR/share. 70% of initial position remaining.
Curevac’s Vaccine doesn’t really work:
https://www.ft.com/content/0309b8ae-bae8-42ce-99e9-d487cd43899c
Good for BioNTech, not so good for European vaccination efforts.
Curevac vaccination plan was badly executed. Pfizer/BioNTech is on the other end of the spectrum and they have been doing beyond expectation so far. Current production target is 3 billion doses in 2021, and more than 3 billion in 2022. Still, consensus earning estimates are rather conservative.
The Economist with an interesting read about India’s privately held Serum Institut, incl. some industry insights (price points)
https://www.economist.com/business/2021/03/03/a-billion-plus-covid-19-shots-in-2021-can-serum-institute-do-it
Good summary. I also followed this stock since a while, and agreed with most of your points.
My quick numbers are that Pfizer/BioNTech can produce up to 2 billion doses in 2021 (about 1.7b confirmed as of now) , and up to 3 billion in 2022. Current price per dose is around 19 usd and a margin of “high 20s”, according to Pfizer. I cross-checked with my own estimates, and in short, let’s say Pfizer/BioNTech will earn about 5 usd profit per dose. After 50/50 split each will earn 5 billion net in 2021, and 7.5 billion net in 2022. There will be a huge R&D cost though since a truck load of vaccines are in the pipeline. These numbers do not include 2023 onward, nor the Fosun’s share of vaccines sold to the greater China region.
You should also post your new articles on Seeking Alpha. We need more analysis on value stocks there. There is a greater number of audience on the platform. Blogging is good but people don’t use feeders to read news or blogposts anymore.
thanks for the comments. Seeking Alpha: No, that is too much work for now.
Another great FT article about Curevac:
https://www.ft.com/content/c3159721-6e64-45eb-8935-22ad8cdf3eb5
Interesting FT article why the “Big 3” vaccine makers missed out on the Covid-19 vaccine and how the vaccine market has become “sexy” again:
https://www.ft.com/content/657b123a-78ba-4fba-b18e-23c07e313331
Genuine question: are these allegations true.
Genuine answer:
This guy Reiner Fuellmich seems to be one of the main people behind the “Querdenker” movement, so anything this guy posts should be not taken seriously.
On a factual level: The vaccine doesn’t of course cure an existing infection and only has limited effect in the first few days after the first shot. So yes, in theory, such a scenario could happen when the virus is already circulating under these unfortunate seniors when they get their first shot.
MMI
Interesting article on how Kariko failed in Academia:
https://www.wbur.org/commonhealth/2021/02/12/brutal-science-system-mrna-pioneer
Memyselfandi007,
I responded a few times but for some reason message won’t post…? Is there a word limit 🙂
Sorry, went into spam…didn’t see it.
Thank you for the interesting analysis. Probably it could be helpful to understand more about the production process of mRNA vaccines (some opern questions above). I found the following article useful:
https://blog.jonasneubert.com/2021/01/10/exploring-the-supply-chain-of-the-pfizer-biontech-and-moderna-covid-19-vaccines/
MMI, I think you’ve lost your marbles!
(couldn’t resist!)
Seriously though, there’s on order of magnitude more unknowns here to be able to have any idea of outcome, and you yourself openly admit you have no edge in this matter – this is just a wild bet. It’s getting close to bitcoin land!
Thanks for the honest feedback. I really appreciate honesty 😉
I agree it is a bet, however, based on a multi-billion EUR profit “proof of concept” and at least a base layer of “real value”.
That’s maybe the main difference to “Bitcoin Land” in my opinion. But I could be wildly wrong of course.
100% agree. I read similar analysis on Reddit.
Thank you very much for the BionTech post from a long time silent reader!
One addition to the risk list is a public campaign that has been kicked off targeting BioNTech employees to leak the vaccine IP. Details can be found here: https://biontech-leaks.org/en/
Thanks for the comment and the link. I am not too surprised by this. However I rely on Dr. Sahin having chosen the right people as employees. I think sooner or later they will sell the vaccine for less than they are currently doing.
I invested in them @95usd because I read a paper where they target autoimmune diseases with an actual therapy (disease-ending). The potential is huge. Of course it’s a huge bet, they are not alone on mRNA tech but I think they will revolutionize modern medicine. I should invest in their competition as well. Disclaimer : have an autoimmune disease, I’m biased
Pak,
thanks for the comment. I think it makes indeed sense to follow the competition closely.
mmi
Agree with most of your analysis of BioNTech, except for the valuation and COVID-19 vaccine potential. While the mRNAs were out first and will certainly capture a decent portion of the market, they will soon start to lose incremental orders for COVID-19 vaccine to Novavax. Their vaccine is easier to produce at scale (the have factories built around the world already), can be stored at refrigeration (vs. colder for mRNA), and has much better tolerability (see AE data for second dose of mRNA). The next phase of the COVID-19 market will be protecting against multiple strains. The mRNA companies plan on doing this with booster shots (that’s fine but tolerability will be an issue). Novavax has proven they can develop vaccine against multiple strains (called a multivalent or bivalent vaccine) through their work on Nanoflu, which itself will be a blockbuster after beating Sanofis $2bn/year flu vaccine in a recent trial. Overall, agree BioNTech and Moderna are the sexier companies (and likely have niche future pipeline in select applications) but Novavax is much cheaper and will come to dominate the COVID-19 market with their bivalent drug and will likely dominate the flu market soon.
Thanks for the comment.
Three points:
– so far it looks that the Novavax vaccine has problems with the SA mutation (only 60% efficacy)
– cooling is not an issue for Moderna and BioNTest indicates that they will have a version for “normal refrigerator temperature” in early summer
– as mentioned, after the “traumatic” experience for Europe from the vaccine distribution, there is a strong desire not to be dependent on a US company when it comes to vaccines. So at least in Europe, Novavax will only have limited chances to sell a lot of vaccine
But let’s see how this works out.
I was wondering do you have a link for the claim of the possibility of elevated temperature vaccines of Moderna/Pfizer?
and one additional question (also to the anonymous biotech investor). I never understood why mRNA is easier (and faster) to manufacture compared to eg. Novavax (who – if I understood correctly – use insect cells). How is mRNA manufactured? Is it done via chemical synthesis or also via cell lines? if the latter – why is it easier to manufacture than eg. Novavax vaccine. Of course mRNA is a far smaller molecule but also more unstable than a protein.
Mao,
for BioNTech, this was mentioned in the Spiegel interview I have linked to:
DER SPIEGEL: Thus far, the vaccine has to be kept at a temperature of minus 70 degrees Celsius during storage and transportation. It became apparent from the very beginning of the vaccination campaign that this can create problems. Will it be possible to adjust the serum in the near future such that it can be stored at normal refrigerator temperatures?
Türeci: The situation will remain unchanged for the next three months, but then we will likely be able to improve the specifications for transportation and storage. Our refined vaccine generation, which will be stable at much higher temperatures, could be available by late summer.
Moderna’s vaccine which is already available can be stored in a normal freezer at -20 degrees celsius and then would last another 30 days in the normal refrigerator:
https://www.npr.org/sections/health-shots/2020/11/17/935563377/why-does-pfizers-covid-19-vaccine-need-to-be-kept-colder-than-antarctica#:~:text=Moderna%20says%20its%20vaccine%20candidate,for%20up%20to%2012%20hours.
With regard to production & Novavax: I am not sure if mRNA vaccines can be produced quicker than the Novavax vaccine. It clearly can be produced quicker than the vector vaccines, where the process from beginning to production seems to require 6-9 months. From what I understand however mRNA vaccines could be “reprogrammed” to mutations if needed within a short period of time. I am not so sure about the Novavax vaccine.
I haven’t deeply researched Novavax, but their disadvantage is clearly that so far they have not gone live and Moderna and BionTech have vaccinated millions of people in the meantime. That’s often called a “first mover advantage”.
MMI
Novavax efficacy is same/better in the original COVID-19 strain (vs. mRNAs) and better in UK strain than Moderna/BioNTech. Neither of the current mRNA ones will have high coverage in the South African (and Brazilian) variants. That’s exactly why they’re developing boosters. Novavax was just the first/only to generate the data in the South African variant.
The Novavax vaccine will also be widely used in Europe (see current rolling EMA review). It’s literally impossible for Europe to have enough of the BioNTech one. EU has already made public they’re negotiating with Moderna, Novavax and others.
Not sure about feasibility of 4C stable mRNA, suspect it’s possible but tolerability will be even worse than now.
Will be interested to see a follow up post in a year or so on performance of NVAX vs. BNTX. Either way, thanks for sharing your perspective.
Anonymous,
yes it will be interesting to compare in a year.
However allow me some more comments as I want to learn more:
– from where do you get your claim that Novavax has better efficacy than BioNTest or Moderna ? I haven’t found any source confirming this, so a link would be really helpful to support your claim and improve the discussion
– Moderna and BioNTech can develop boosters because it is an easy thing to do for them and only takes them a few weeks. Can Novavax adjust its vaccine as easily or are they more restricted like traditional vaccine companies which need 6-9 months to change their vaccine ?
– Yes, Novavax will be used in Europe in 2021. the question is: What is there after ? BioNTech is massively scaling production and my assumption is that Novavax will be less important in 2022 at least in Europe unless it would be indeed much better
– What do you mean with “tolerability will be even worse than now” ? So far, very few side effects have been reported, despite millions of vaccinated people both, for BioNTech and Moderna. In Germany there was a case where some people mistakenly got 5-6 times the dose they should have received without any side effects. So again, a source for your claim would be highly appreciated.
Sure – happy to share.
Efficacy
So when I say it’s “better/comparable”, I mean numerically better. As always, it’s extremely difficult to do cross-trial comparison but to the best of our abilities, they appear comparable (or the raw numbers are better for Novavax b/c trial population is older). The important thing to do is do an apples-to-apples comparison since there have been many variants that have evolved since the pandemic evolved. Novavax headline number was a combination of “original” nSARS-CoV2 strain and UK 501Y.V1 variant which showed the ~89% efficacy. Both mRNA drugs also have shown a decrease in efficacy in the UK strain as well (worse than Novavx). In the “original” nSARS-CoV2 strain we have the following data:
Novavax’s NVX-CoV2373: 95.6% (see Jan 28th press release & slide 10 of investor deck here: https://ir.novavax.com/static-files/2f6f14cb-3205-4719-b28c-1711793b9782)
Moderna’s mRNA-1273: 94.1% (see NEJM publication: https://www.nejm.org/doi/full/10.1056/NEJMoa2035389 or press release: https://investors.modernatx.com/news-releases/news-release-details/moderna-announces-primary-efficacy-analysis-phase-3-cove-study)
Pfizer/BioNTech’s BNT162b2: 95% (see NEJM publication: https://www.nejm.org/doi/full/10.1056/NEJMoa2034577 or press release: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-conclude-phase-3-study-covid-19-vaccine)
Safety
The same caveats as above about cross-trial comparison apply here. It’s difficult to do and is imprecise. Based on the data we have the Novavax vaccine has significantly better tolerability than the mRNA vaccines (this is not surprising but has implications about future success). For grade 3 or above adverse events (grade 3 is considered “severe”, anything above is life-threatening or death). At grade 3, a person is unable to do “tasks of daily living” (i.e. bed-ridden), this is what we have:
Novavax’s NVX-CoV2373: we have any severe adverse events rate of 1.1% (I haven’t seen the detailed breakdown yet but the phase 1 data support this. Source is slide 24 of deck: https://www.novavax.com/sites/default/files/2021-02/20210202-NYAS-Novavax-Final.pdf)
Moderna’s mRNA-1273: we have any severe systemic adverse event rate of 17.1% after second dose (it’s mostly fever, arthralgia, myalgia, fatigue, headache. Source is table 3 here: https://www.cdc.gov/vaccines/covid-19/info-by-product/moderna/reactogenicity.html. The trail data also support this)
Pfizer/BioNTech’s BNT162b2: we have grade 3 fatigue at 4.6%, grade 3 headache of 3.2%, grade 3 mylagia of 2.2% (source is table 3 from here: https://www.cdc.gov/vaccines/covid-19/info-by-product/pfizer/reactogenicity.html. Trial results corroborate on slides 17 & 18: https://s21.q4cdn.com/317678438/files/doc_presentations/2020/09/Covid-19-Programs_FINAL.pdf)
Boosters vs. Multivalent to Address Variants
Also too complicated to explain in brief but will summarize. Basically the mRNAs will likely be limited by dose size on being able to address multiple variants. Accordingly, they will have to make individual “boosters” for each new strain/variant. That’s fine and certainly better than having nothing but will run into supply chain and dosing issues (i.e. dosing the right people to protect against the right variant in sufficient volume. The problem gets worse because of the reactogenicity of the mRNAs – the more doses you give the more side effects people have so if you’re giving people vaccines for 4-5 (or more) variants you can imagine lots of adverse events). The way the developed world treats seasonal flu is through polyvalent/multivalent vaccines that address multiple strains at the same time so you don’t have to worry about matching strains to people and giving everyone shots against 5-10 (or more) different strains. Novavax is developing exactly that (and they have what appears to be the best multivalent vaccine against seasonal flu though there are others out there as well). nSARS-CoV-2 will become exactly like the seasonal flu. We will all receive annual (cadence may be slightly different) vaccines against multiple different strains until either the virus completely disappears (unlikely) or it becomes permanently endemic (appears it already has). As for timing: all three companies (Pfizer/BioNTech, Moderna, Novavax) have announced that they’re developing pre-clinical constructs for their booster or bivalent vaccines. None are in the clinic and all plan to be in the clinic in Q2 2021. To your question on timing: Novavax should be able to develop new multivalent ones in roughly same time as an mRNA strategy but hard to say as depends on many factors.
Novavax will be used widely in the future not even so much because it’s better (which it is) but because a multivalent approach makes much more sense than giving 5-10 shots to each person. The market will play out very similarly to how the annual flu vaccine market does. The reason we don’t have an annual mRNA flu vaccine is not so much because the technology is new but because the tolerability is poor (vs. alternatives) for a vaccine everyone should get and because the handling/manufacturing capacity, including cold-chain is more expensive. At this point, it’s an inferior technology for vaccination though I suppose that could change. We just have no evidence (that I’m aware of) that suggests mRNA approach will be better.
I won’t get into the manufacturing because it’s much more difficult to measure but suffice it to say that manufacturing a nanoparticle protein is easier than making liposomal mRNA. All companies will have capacity for ~2 billion doses per year but the mRNA companies will likely face shortages of liposome ingredients (potentially others too). They’re also more expensive to make to begin with (for confirmation: see public comments from Pfizer vs. others about COGS).
Thanks for the comment. I’ll need to digest that. However I see one BIG advantage forPfizer/BioNTech: They have now “live study” of 8 mn people with real time data: Israel.
https://www.timesofisrael.com/vaccine-found-92-effective-in-israel-in-first-controlled-result-outside-trials/
Assuming that there are also mutant virus infections in Israel, 92% efficacy on 166000 people (!!!) looks impressive.
Maybe one remark to the flue shots: As mentioned: “Multivalent” flu shots are standard because by the time you produce the vaccine, you do not know how the flu virus will actually look like. So my maybe naive assumption is that you don’t need this “start shots” in the future if you can actually produce the vaccine against the relevant virus within short time.
But thanks for the detailed contribution !!!
Anon, can I just say a huge thanks for taking the time to write your informative comments. Very interesting g.
On the temperature subject Arcturus CEO was saying that their vaccine will be liphilised (i.e. in powder, which can be mixed with serum before administration), so in that case temperature will no longer be a factor. Also the CEO of Moderna has said that he expects that the temperature requirement for preservation of his vaccine (currently -20ºC) will be much less stringent over the next 18-24 months, they are working on it.
Very interesting. Coincidentally I spent part of my day starting to look at the space. Modena seem to be the gold standard, probably followed by Biontech. There is also Curevac and at least two small listed US companies (Transfer Bio and Arcturus).
The potential for the space is phenomenal, it could disrupt healthcare forever. Valuations are very lofty. I agree with your approach is starting a small position. If a second indication is approved things are likely to start moving much faster. I would not lose sight of Merck and Sartorius as key suppliers to the industry.
There is a very good explanatory podcast about mRNA from Ark Invest, interviewing the CEOs of Modena and Arcturus.
https://ark-invest.com/podcast/understanding-mrna-conversations-with-moderna-and-arcturus-therapeutics/
Best
Thank you. This is extremely interesting stuff !!
SUPER Bericht!
Viel davon gelernt, ganz tolle Zusammenfassung und echt horizonterweiternd.
Which reminds me, I should really introduce your website in my next “Blogs to Watch” series. In your particular case, I’ll send you my article in advance so that you can check if you are happy with what I write. Will be a few weeks as I have another series coming up, but I’ll touch base with you about it.
>
Here is one confirmed case of a successful sign up because of “blogs to watch”. Thanks guys – I enjoy reading articles by thorough German investment authors like you 🙂
Welcome !!