Contingent Value Rights (CVRs) – The case of the Sanofi/Genzyme CVR
As I was trying to research a little bit how to value a pipeline of drugs still in development (Actelion spin-off), I stumbled across the so-called “Contingent Value Rights” (CVRs) which are often used in Pharma takeovers.
A CVR is somehow similar to a tracking stock with the exception that the CVR often tracks a more specific item such as a single product or in case of many Pharma M&A transactions, the outcome of a certain drug development project.
Acquirers and sellers sometimes use this instrument if they cannot agree on the value of an under development drug. The idea behind is that the seller keeps the upside and the buyer doesn’t need to pay upfront for some very risky future cashflows.
Sanofi/Genzyme Lemtrada CVR
When Sanofi took over Gynzme in 2011 such a situation crystalized. This is from a 2015 NYT story:
In 2011, Sanofi acquired Genzyme for $20 billion. At the time, Genzyme was conducting clinical trials of a promising multiple sclerosis treatment called Lemtrada.
Genzyme’s shareholders didn’t think Sanofi’s $20 billion offer reflected Lemtrada’s upside. So Sanofi agreed to issue one contingent value right for every Genzyme share in the buyout. The rights gave their holders an additional $3.8 billion if Lemtrada cleared certain hurdles. These included gaining approval by the Food and Drug Administration by March 31, 2014, and reaching specific sales targets within certain time frames.
At the time of the merger, Genzyme estimated that each contingent value right was worth $5.58. It recommended shareholders approve the deal, and they did.
The CVR was issued as a traded instrument under the ticker GCVRZ. For those who are interested, there is more info on the drug and the CVR in a 2011 post and a 2014 post on Value Investor’s Club which were both quite bullish.
As we can see in the chart, after increasing initially to a value of~2,60 USD per CVR, over time the value steadily decreased with a bottom in 2015 of around 0,08 USD per CVR in March 2016 (according to Bloomberg there are 236,4 mn CVRs outstanding):
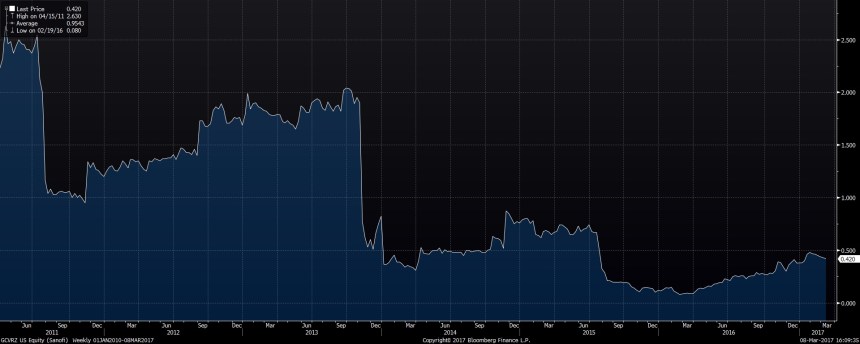
So what happened ? Did the drug not work ? Well it seems to be more complicated:
Under the agreement, Sanofi would make “diligent efforts” to shepherd Lemtrada through the F.D.A. approval process and promote it as it would any drug. This set out a higher standard than Sanofi would have faced under an agreement to make only a “reasonable effort” with the drug.
The agreement also required Sanofi to “ignore any cost of potential milestone payments in working to gain regulatory approval and commercialize Lemtrada,” according to a lawsuit filed on Nov. 9 in federal court in New York.
Here is where the conflict comes in. During this period, Sanofi was developing its own multiple sclerosis drug, Aubagio, which would compete with Lemtrada.
Sanofi, therefore, had a choice. It could do what it had promised Genzyme’s shareholders: work diligently to secure F.D.A. approval of Lemtrada and market the drug. But success in these efforts had a downside: the additional $3.8 billion in payments to Genzyme rights holders.
Aubagio, by contrast, had no such downside. If Sanofi focused on developing and promoting Aubagio, it would generate profits that required no expensive payments to rights holders.
So the drug underlying the CVR was doing OK but Sanofi had no incentive to let this drug succeed because they had a competing drug where they could keep 100% of the profits.
As this is the US, of course a lawsuit was filed and Sanofi is on the hook for a potential large payout:
In the complaint, the American Stock Transfer & Trust Company, the trustee representing the Genzyme rights holders, contended that Sanofi failed to fulfill its obligations under the deal. As a result, investors have not received at least $708 million they were owed, the suit said.
According to this update from early 2017, at least one of the law suits has been dismissed.
Sales targets & Option values
Unfortunately, the official reporting of Sanofi is not really clear about how the overall picture looks like. It seems that the first milestone mentioned in the 2014 VIC post2014 VIC post has not been met.
Also the next milestones seem hard to reach. If I read the report correctly, current sales are ~500 mn USD per year and until 2020 this would need to more than triple in order to reach the 1.8 bn annual sales required.
Without deep research it is really hard to deduct any option value here. I am also not sure if there is still a pending lawsuit which would have some option value in this case.
Overall, the CVR clearly seems to be something where a real specialist either in MS drugs or litigation could add value but not a private value investor.
Nevertheless the current CVR shareholder base looks like a “who is who” of “special situation hedge funds. Here are the top 10 holders:
STONEHILL CAPITAL MANAGEMENT INC STO 13F 32,062,445 13.56 10,332,843
ELLIOTT MANAGEMENT CORPORATION ELL 13F 20,454,561 8.65 0 12/31/16
MASON CAPITAL MANAGEMENT LLC MAS 13F 14,644,398 6.19 0 12/31/16
TPG GROUP HOLDINGS (SBS) ADVISOR TPG 13F 14,420,462 6.10 -5,820,000 1
BRIGADE CAPITAL MANAGEMENT LP BRI 13F 12,268,815 5.19 52,984
WHITEBOX ADVISORS LLC WHI 13F 11,189,907 4.73 -4,970,542 12/31/16
POINTSTATE CAPITAL POI 13F 7,305,164 3.09 7,305,164 12/31/16
FARALLON CAPITAL MANAGEMENT LLC ULT-AGG 6,827,805 2.89
GRUSS CAPITAL MANAGEMENT GRU 13F 5,500,000 2.33 0 12/31/16
MICHAEL F. PRICE MIC 13F 3,583,668 1.52 0 12/31/16
So some of these guys seem to think that there is value but they also have the money to pay specialists and big league lawyers.
Link to the Actelion case
What I found really interesting as a learning experience is the fact, that there really can be huge conflicts of interest between the CVR holders and the company, which in the Lemtrada case looks quite suspicious.
I guess this is one of the reasons why the Actelion CEO insisted on the Spin-off instead of CVRs. In a well-funded spin-off company he has the whole development and licensing process under control and can work to the benefit of the spin-off shareholders without relying on the buyer.
Summary:
The Sanofi CVR is an intellectually interesting case but without specialist knowledge clearly a case for the “too hard” pile. For investors, the spin-off structure like in the Actelion case has clear benefits compared to the CVR structure.
Hi, do you know if the BMY/Celgene CVR trades? Have you had a look at it?
any updates on where this is going?
The legal motion will fail as the underlying premise is wrong (cause of intent)
Alemtuzumab (Campath/Lemtrada) is a high efficacy pulsed immune reconstruction compound. Unlike Aubagio that is a medium efficacy maintenance compound.
MS is not 1 disease: there are different types of MS and different treatment with different risk profiles to depending on the prospect a patient is facing at a given point in time.
I (me and myself) participated in the Campath clinical trial 3b at Cambridge, and was an unfortunate CVR holder.
Alemtuzumab is in the same risk/reward category as BIIB’s Natalizumab and NOT AUBAGIO.
As such, the cause of intent will be dismissed. I am now short the CVR.
To complicate matters further, I think (i can’t find the link) that Sanofi bought a large chunk of the GCVRZ around 1.60, only just before the big drop in fall of 2013. Whether or not a judge will find that significant, it does imply that Sanofi was aware of the possibility of Lemtrada success and wanted to hedge its bets a bit on paying out the CVR holders….
That was a dutch auction tender in September 2012 …
Click to access 31076_20120904_GENZYME_en.pdf
Interesting…